Trading Update
08/06/21
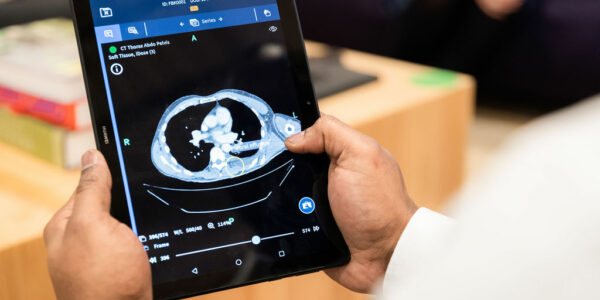
Our trading during the last period has met management expectations. We have secured our first commercial contract for our flagship product Bleepa. During the period, we have continued to develop Bleepa to include key features such as photo-capture which has led directly to the award of this commercial contract. We have also developed capabilities in cloud hosting to enable more cost-effective delivery and scalability.
Recognition
Importantly, Bleepa became the only clinical communication platform to be appointed to the national NHSx Clinical Communications Framework with a CE mark as a Class 1 Medical Device for medical imaging display.
During the year Feedback Medical Ltd. was successfully re-certified as a medical device manufacturer with ISO 13485 accreditation for its manufacturing processes. Additionally, achieving ISO 27001 accreditation for data security and data management. We have also received a Cyber Essential Plus accreditation for cyber security and DCB0129 clinical safety and risk management certification. The emphasis on quality and regulatory compliance is a clear differentiator for us and our products.
COVID-19
2020 has been a difficult year for the healthcare sector with many of our customers being overwhelmed by COVID-19. On one hand, COVID-19 has strongly demonstrated the need for our leading technology. However, it has also made it very difficult to engage customers who were understandably focused on, initially, treating acute patients and, subsequently, implementing the national vaccine program. We are now seeing increased interest for Bleepa as NHS organisations emerge from the acute COVID-19 response and begin to focus on solutions that can help them address the growing backlog of elective and cancer care cases. Additionally, solutions that can facilitate more robust communications ahead of any further waves of COVID-19. Bleepa is now well-positioned to recognise this increasing customer interest in its domestic market.
The performance of Bleepa during the pandemic is also beginning to be recognised in international markets and has been supported by recent DIT trade missions to India. We are actively responding to this interest and have invested in a presence on the ground in India to help convert a growing pipeline of opportunities, although these efforts have recently slowed due to the recent surge of COVID-19 in India.
Revenue
Our balance sheet remains strong, and unaudited net cash as of 31 May 2021 was £2.2million (31 May 2020: £0.7 million). We expect unaudited revenue for the financial year ending 31 May 2021 to be approximately £0.28m (FY20: £0.45 million). The reduction in revenue stems from the realisation of the previously anticipated decline in TexRAD sales which formed the basis of the investment in Bleepa and the pivot in corporate strategy in 2019. Importantly we have now started to recognise revenue from its new Bleepa product line in the current financial year.
Although the total number of sales has declined, based largely on TexRAD, the move to Bleepa has also seen the average contract value increase, generating better margins per sale, this will enable the company to drive profitability as well as revenue going forward. The company has already booked £0.12m of revenue to be recognised in the coming financial year and expects revenues to grow as sales of Bleepa increase in the year ahead.
Outlook
We remain focused on increasing revenue-generating opportunities and addressing a wide range of markets both domestically and internationally for Bleepa. While it continues to be a core target, management is excited by the increasing number of prospects and leads both in and outside of the NHS and has a strong pipeline of opportunity. There is a growing requirement for remote solutions and Bleepa leads at the forefront of the market. Management expects to benefit from the increasing demand for its solutions enabling it to grow repeat revenues and benefit from increased visibility afforded by its Software as a Service model.