Safe, secure and compliant clinical messaging
19/05/21
Safe, secure and compliant clinical messaging: lessons from the COVID-19 pandemic
Feedback Medical Limited hosted a Digital Health webinar on Wednesday 12 May at 12pm focusing on safe, secure, and compliant clinical messaging. This also included exploring some of the lessons learned from the COVID-19 pandemic.
The panel discussion, chaired by Digital Health CEO and Editor-in-Chief Jon Hoeksma, featured Feedback Medical Ltd CEO Dr. Tom Oakley, Pennine Acute Hospitals NHS Trust Chief Clinical Information Officer and Consultant Chest Physician Georges Ng Man Kwong, and Beachcroft LLP Senior Associate Darryn Hale.
Watch the full Bleepa webinar
The importance of a CE marked medical device
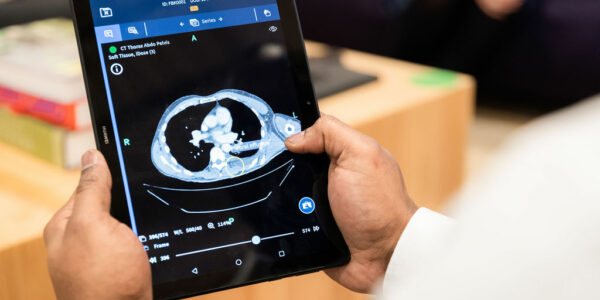
Following introductions from the panel, the webinar focused on the importance of any platform that ‘displays digital patient images for the purpose of diagnosis’ used by clinicians. The panel discussed how platforms such as these should hold a CE mark as a regulated medical device. Bleepa is the only clinical messaging app holding a CE mark on the NHSX clinical communications framework.
Role of organisations as data controllers under GDPR
Under GDPR obligations, patient data created in the course of an employee’s work is the responsibility of the employing trust as a data controller. Therefore, this raises concerns about meeting these obligations effectively when clinicians are using mainstream messaging apps for patient case discussion:
- Data processing agreements – As data controller, data processing agreements must be in place with all products that are used by clinicians. This applies even where these are free or used without the Trust’s express permission.
- Active prevention – Steps must be taken to show how trusts have actively prevented the use of free apps. Such as, engaging with a commercial alternative, which is the simplest way to do this.
Furthermore, any clinical discussion in mainstream messaging apps is not recorded within the patient record. This raises concerns not only about GDPR obligations but also potential patient safety issues.
Engagement from delegates
A plethora of great questions from delegates covered broad topics, such as:
- The benefits of using Bleepa as opposed to a PACS viewer
- Integrating with EPRs and other clinical systems
- The scope of Bleepa’s use beyond secondary care
- How it can be applied to multi-disciplinary team meetings, both synchronously and asynchronously
Highlights
Watch the highlights, focusing on the data security and regulatory considerations for clinical communication via messaging apps.
Find out more about the Digital Health webinar here.