Bleepa affixed with a UK Medical Device Certification
29/06/21
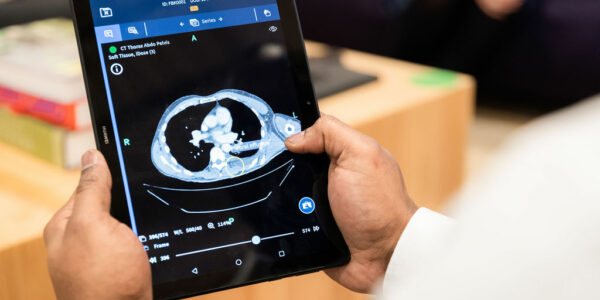
We are happy to announce that Bleepa has gained a UK Medical Device Certification (UKCA) mark.
UK Medical Device Certification
The UKCA is the post-BREXIT UK regulatory certification and has been affixed to Bleepa. Bleepa is our flagship medical imaging communications platform. This new certification adds to our portfolio of regulatory approvals for the tool, further validating Bleepa’s technology.
“In addition to the CE mark affixed last year, Bleepa has now enhanced its regulatory technical file to include the UK’s medical Device Manufacturing mark. The new UK product marking is used for goods being placed on the market in Great Britain post-BREXIT. It further validates as well as provides confidence in the quality and expertise of Bleepa’s technology.” Dr Tom Oakley, CEO of Feedback